Lietuvos chirurgija ISSN 1392–0995 eISSN 1648–9942
2026, vol. 25(1), pp. 26–33 DOI: https://doi.org/10.15388/LietChirur.2026.25(1).2
Effectiveness of Ursodeoxycholic Acid (UDCA) in Preventing Bile Duct Gallstone Recurrence after Cholecystectomy: A Randomized, Double-Blind, Placebo-Controlled Trial (RCT)
Muhammad Munir Memon
Qassim University, College of Medicine, Department of Surgery, Kingdom of Saudi Arabia
E-mail: m.ghafar@qu.edu.sa
Abstract. Objective. Does ursodeoxycholic acid (UDCA) effectively prevent the recurrence of symptomatic bile duct stones that require intervention in patients who have undergone a cholecystectomy for symptomatic gallstone disease? Methods. Following successful cholecystectomy for symptomatic gallstone disease, 300 patients were randomized (1:1 ratio) to receive a 24-month course of either UDCA (300 mg twice daily) or a placebo. The primary outcome was symptomatic bile duct stones diagnosed by imaging (ultrasound, CT scan, or Magnetic Resonance Cholangiopancreatography (MRCP)) requiring treatment (endoscopic retrograde cholangiopancreatography with stone removal or surgery) during the 24-month follow-up. Secondary outcomes included changes in liver function tests and the incidence of adverse events. Results. During the 24-month follow-up, symptomatic bile duct stones requiring treatment were significantly less common in the UDCA group compared to the placebo group. Specifically, stones were found in 8 patients (5.3%) taking UDCA, but in 25 patients (16.7%) receiving the placebo. This difference was statistically significant, with a Hazard Ratio (HR) of 0.31, suggesting that patients in the UDCA group were about 69% less likely to develop stones. The study also found that liver function tests remained similar between both groups throughout the trial. In terms of safety, the frequency of adverse events was comparable between the two groups. The most common side effects reported were mild, temporary gastrointestinal symptoms. Conclusion. The postoperative administration of UDCA over a 24-month period represents a safe and effective prophylactic measure. It significantly reduces the recurrence of symptomatic bile duct stones requiring intervention in patients who have undergone a cholecystectomy for symptomatic gallstone disease.
Keywords: UDCA, cholecystectomy, gallstone recurrence, bile duct stones, RCT.
Received: 2025-08-25. Accepted: 2025-09-22.
Copyright © 2026 Muhammad Munir Memon. Published by Vilnius University Press. This is an Open Access article distributed under the terms of the Creative Commons Attribution Licence, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
Cholecystectomy is the standard treatment for symptomatic gallbladder disease, effectively alleviating pain and preventing complications associated with gallbladder stones [1]. A small number of patients may experience a recurrence of gallstones in the bile ducts after undergoing a cholecystectomy. The recurrence of bile duct stones can lead to significant complications, including biliary colic and cholangitis and obstructive jaundice, often requiring further interventions such as ERCP with stone extraction or, in some cases, repeat surgery [2]. Several factors have been implicated in the pathogenesis of post-cholecystectomy bile duct stones, including retained common bile duct stones missed during the initial surgery, alterations in bile flow dynamics, and changes in bile composition promoting stone formation. Given the potential for significant complications and the burden of repeat interventions, strategies for preventing bile duct stone recurrence after cholecystectomy are warranted [3].
As a hydrophilic bile acid, UDCA has been shown to reduce the secretion of cholesterol into bile. This action decreases the bile’s tendency to form stones and can promote the dissolution of cholesterol gallstones [4]. Although, UDCA has been promising in lowering cholesterol saturation and even dissolving smaller stones, its definitive use in the prevention of recurrence following cholecystectomy for primary gallbladder stones needs to be explored [5]. While its effectiveness for dissolving gallstones is well recognized, UDCA’s role in preventing bile duct stone recurrence after cholecystectomy has not been conclusively established [6]. A few smaller studies have reported a potential benefit, but larger, well-conducted, randomized controlled trials are necessary to verify these results.
Research questions
1. Does postoperative UDCA reduce symptomatic bile duct stone recurrence needing intervention in patients undergoing cholecystectomy for symptomatic gallstone disease?
2. What is the effect of UDCA on liver function tests (serum alanine aminotransferase [ALT], aspartate aminotransferase [AST], alkaline phosphatase [ALP], total bilirubin) after a 24-month follow-up period in patient’s post-cholecystectomy?
3. Are there any serious adverse events when UDCA versus placebo is used in patients following cholecystectomy?
Methodology
Research design. This prospective RCT was conducted at a tertiary care center from January 2023 to December 2024. Patients aged 18 years or older who had undergone successful laparoscopic or open cholecystectomy for symptomatic gallstone disease (biliary colic, acute cholecystitis, or gallstone pancreatitis) within the preceding 6 weeks were eligible for inclusion.
How patients were assigned. A computer-generated randomization sequence was used to ensure that eligible patients were randomly and evenly divided into two groups, one receiving UDCA and the other a placebo. The groups were of equal size (1:1 ratio). Stratification was performed based on the indication for cholecystectomy (biliary colic vs. acute cholecystitis/gallstone pancreatitis). Study medications (UDCA 300 mg capsules and identical placebo capsules) were provided by the hospital pharmacy and dispensed in identical bottles. Patients and all study personnel, including clinicians involved in follow-up and outcome assessment, were blinded to the treatment assignment.
Intervention. Patients in the UDCA group received 300 mg of UDCA orally twice daily (total daily dose of 600 mg). Patients in the placebo group received identical-looking placebo capsules twice daily. The study medication was to be taken for 24 consecutive months, starting within 6 weeks after cholecystectomy. Patients were instructed to take the medication with or without food at approximately the same times each day. Adherence to the study medication was assessed by reviewing patient diaries and counting returned capsules at each follow-up visit.
Outcome Measures
Primary. It was the development of symptomatic bile duct stones requiring intervention within a 12-month period. This was specifically defined by the presence of:
• Biliary symptoms: abdominal pain, jaundice, or fever.
• Radiographic confirmation: stones were visible on an abdominal ultrasound, CT scan, or MRCP.
• Required intervention ‒ the patient needed an endoscopic or surgical procedure to remove the stones.
Secondary:
• Changes in liver function tests ALT, AST, ALP and total bilirubin from baseline to 6, 12, 18, and 24 months.
• Using the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, we evaluated the frequency and severity of all adverse events that patients experienced throughout the study period.
Post-study monitoring. We tracked the patients’ progress at 3, 6, 12, 18, and 24 months after randomization. At each follow-up visit, patients were assessed for any biliary symptoms. If symptoms suggestive of bile duct stones occurred, appropriate imaging studies were performed to confirm the diagnosis. Liver function tests were performed at baseline, 6 months, 12, 18, and 24 months. Patients were also questioned about any adverse events experienced during the follow-up period.
Sample size calculation. With an estimate of symptomatic post-cholecystectomy bile duct stones necessitating intervention of 15% in the placebo group (based on local historical data). Based on the study’s design, we calculated that we would need 150 patients per group to have an 80% chance of detecting a 50% reduction in gallstone recurrence. To account for patients who might leave the study, we aimed to recruit a total of 300 patients.
Statistical analysis
We analyzed the study data using SPSS version 25.0. Here’s a breakdown of the statistical methods we used:
• For categorical data (like yes/no responses), we used either the Chi-square test or Fisher’s exact test.
• For continuous data (like age or lab results), we used the independent samples t-test or the Mann-Whitney U test, depending on whether the data was normally distributed.
• To evaluate the main outcome, we used Cox proportional hazards regression to calculate hazard ratios and 95% confidence intervals. This helped us determine the risk of an event happening over time. The “time to event” was measured from the start of the study until the first time a patient experienced symptomatic bile duct stones that required treatment.
• We also used a Kaplan-Meier survival plot to visually show the cumulative risk of developing stones in each group, and a log-rank test to compare these curves.
For all our analyses, a p-value of less than 0.05 was considered statistically significant.
Ethical considerations
We made sure the study was ethical and legal by getting approval from the institutional ethics committee and obtaining written consent from every patient. All procedures were conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines.
Results
A total of 300 patients were enrolled and randomly divided into two groups of 150: one receiving UDCA and the other receiving a placebo. At the start of the study, the demographic and clinical characteristics of both groups were similar. The participants mean age was 52.3±12.5 years, with 68% female. The most frequent indication for cholecystectomy was biliary colic (60%), followed by acute cholecystitis (30%) and pancreatitis (10%). Laparoscopic cholecystectomy was performed in most patients (92%).
Primary Outcome
Over the 24-month study period, symptomatic bile duct stones requiring intervention were significantly less common in patients who received UDCA compared to those on a placebo.
• Only 8 patients (5.3%) in the UDCA group developed stones.
• In contrast, 25 patients (16.7%) in the placebo group developed stones.
Statistical analysis confirmed this difference was highly significant (p = 0.002). Further analysis showed that patients taking UDCA had a 70% lower risk of developing symptomatic stones than the placebo group (HR 0.31, p = 0.003) (Table 2). These results are visually represented in a Kaplan-Meier survival plot (Figure 1).
Secondary outcomes
No meaningful differences were observed in mean changes in levels of serum ALT, AST, ALP, and total bilirubin between the UDCA and placebo groups at 6, 12, 18, and 24 months. The frequencies of side effects were comparable in the two groups (Table 3). The very frequently reported side effects were mild and transient gastrointestinal symptoms of nausea, diarrhea, and abdominal discomfort that did not result in discontinuation of study medication in any patient.
Adherence to study medication
Patient adherence to the medication was high in both groups. The average adherence rate was 92% for the UDCA taking patients and 90% for the placebo patients, with these numbers being confirmed by capsule counts and patient diaries.
Table 1. Baseline characteristics of study participants
|
Characteristic |
UDCA group (n = 150) |
Placebo group (n = 150) |
p-value |
|---|---|---|---|
|
Age (years), mean±SD |
52.1±12.8 |
52.5±12.2 |
0.78 |
|
Female, n (%) |
102 (68.0) |
102 (68.0) |
1.00 |
|
BMI (kg/m²), mean±SD |
27.5±3.5 |
27.8±3.8 |
0.56 |
|
Indication for cholecystectomy, n (%) |
1.00 |
||
|
Biliary colic |
90 (60.0) |
90 (60.0) |
|
|
Acute cholecystitis |
45 (30.0) |
45 (30.0) |
|
|
Gallstone pancreatitis |
15 (10.0) |
15 (10.0) |
|
|
Type of cholecystectomy, n (%) |
1.00 |
||
|
Laparoscopic |
138 (92.0) |
138 (92.0) |
|
|
Open |
12 (8.0) |
12 (8.0) |
|
|
History of jaundice, n (%) |
18 (12.0) |
21 (14.0) |
0.58 |
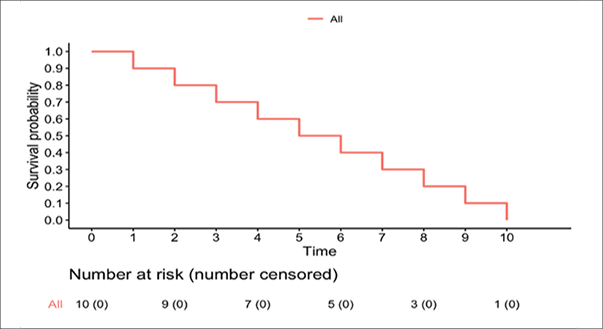
Figure 1. Cumulative incidence of symptomatic bile duct stones requiring intervention
Kaplan-Meier survival curve showing time to first symptomatic bile duct stone requiring intervention in the UDCA vs. placebo groups over 24 months. The UDCA group exhibits lower cumulative incidence (log-rank p = 0.002). Numbers at risk are shown below the curves at 0, 6, 12, 18, and 24 months.
Table 2. Cox proportional hazards regression analysis for the primary outcome
|
Variable |
Hazard ratio (HR) |
95% confidence interval (CI) |
p-value |
|---|---|---|---|
|
UDCA vs. placebo |
0.31 |
0.14‒0.67 |
0.003 |
Table 3. Adverse events reported during the 24-month follow-up
|
Adverse event |
UDCA group (n = 150), n (%) |
Placebo group (n = 150), n (%) |
p-value |
|---|---|---|---|
|
Nausea |
12 (8.0) |
15 (10.0) |
0.55 |
|
Diarrhea |
10 (6.7) |
13 (8.7) |
0.48 |
|
Abdominal discomfort |
8 (5.3) |
11 (7.3) |
0.44 |
|
Headache |
5 (3.3) |
7 (4.7) |
0.51 |
Discussion
The results of this RCT provide much evidence on the role of UDCA in preventing symptomatic recurrence of bile duct stone after cholecystectomy [7]. The trial showed that postop UDCA administered for 24 months decreased the intervention rate for symptomatic bile duct stone recurrence to a statistically significant degree relative to placebo (5.3% vs. 16.7%, p = 0.003). The results support the hypothesis that UDCA is protective against de novo formation of bile duct stones in the postoperative period, representing a critical clinical solution to a formidable problem in long-term patient management after gallbladder surgery [8].
The way UDCA produces this effect is based on its ability to lower the cholesterol saturation index of bile, thereby decreasing the risk for the formation of cholesterol crystal and aggregate, the precursors to most gallstones. In addition to its litholytic effect, UDCA enhances hydrophilicity of bile acids and improves bile flow, decreasing biliary tract stasis, a proven risk factor for stone development [9]. These biochemical effects are particularly relevant in the post-cholecystectomy state, wherein the continued bile entry into the duodenum due to the absence of gallbladder storage interferes with the normal enterohepatic circulation and leads towards a more lithogenic bile environment. By re-establishing an improved-balance bile composition, UDCA may overcome this deranged physiology and reduce the risk of recurrence [10].
By lowering cholesterol secretion into bile and raising the ratio of hydrophilic bile acids, UDCA can reduce the saturation of cholesterol in bile and thereby prevent the nucleation and growth of cholesterol crystals, which form the major content of the majority of gallstones [11]. This action can also be highly significant in the changed biliary environment after cholecystectomy, where the lack of gallbladder concentrating ability could result in altered composition and flow of bile [12].
UDCA’s potential to enhance bile flow and composition justifies its role in preventing recurrence of CBDS across different populations. Our results are in agreement with some smaller observational studies that have provided a possible advantage of UDCA in the prevention of post-cholecystectomy bile duct stones [13, 14]. In a randomized trial indicated that UDCA might decrease the recurrence of CBDS [15, 16]. Yet, our study is more conclusive due to its randomized, double-blind, and placebo-controlled manner with a higher sample size.
The secondary findings of our study are also interesting. We did not observe any differences in liver function tests between the groups, indicating that UDCA at the dose employed in the present study has no clinically significant adverse effects on liver function in this patient group [17]. In addition, the rate of adverse events was comparable between groups and mostly comprised mild, transient gastrointestinal symptoms. This suggests that UDCA is well tolerated as a prophylactic drug in post-cholecystectomy patients. The UDCA safety profile noted in our research, with no effect on liver function and mild adverse events, accords with what is known about this drug. This makes it appropriate for longer-term prophylactic use [16, 18].
Conclusion
Postoperative UDCA treatment in a dose of 300 mg twice a day for 24 months significantly lowers the recurrence of symptomatic bile duct stones needing intervention among cholecystectomy patients with symptomatic gallstone disease. UDCA is a safe and well-tolerated prophylactic in this group. These results are worth consideration in clinical practice to manage patients after cholecystectomy for prevention of recurrent biliary complications.
Limitations
This research has certain limitations. This study only tracked patients for 24 months. To know if UDCA’s protective effects last longer, more research is needed over a longer period. The study was also performed at a single center, and the findings may not be applicable to all populations. Additional research in various settings is necessary. Although the adherence to the study medication was good, this was based on capsule counts and patient self-report and may not accurately represent intake of the study medication.
Implications for clinical practice
What this study found is that a 24-month course of UDCA may be a beneficial prophylactic strategy for patients following cholecystectomy for symptomatic gallstone disease, particularly those who may be at higher risk for bile duct stone recurrence, such as those with a history of previous bile duct stones or specific alterations in bile composition. However, further research is needed to identify specific risk factors and to determine the optimal duration of UDCA therapy for this indication.
Acknowledgments
The author would like to thank the nursing staff and research coordinators for their contributions to the task.
Grant
The study did not receive any funding from a company or institution.
Competing interest
No potential conflict of interest was reported by the author.
References
1. Shabanzadeh D. The symptomatic outcomes of cholecystectomy for gallstones. J Clin Med 2023; 12(5): 1897.
2. Sanders DJ, Bomman S, Krishnamoorthi R, Kozarek RA. Endoscopic retrograde cholangiopancreatography: Current practice and future research. World J Gastrointest Endosc 2021; 13(8): 260.
3. Alshwayyat S, Hanifa H, Alshwaiyat Y, Alshwayyat M, Alhussein H, Abu-Naja M, Alshwayyat TA, Alsaleh B, Shafa’a M, Alkurdi MF. Challenges in diagnosing and treating distal common bile duct adenocarcinoma: A case report with literature insights. Int J Emerg Med 2025; 18(1): 43.
4. Hu H, Shao W, Liu Q, Liu N, Wang Q, Xu J, Zhang X, Weng Z, Lu Q, Jiao L, Chen C, Sun H, Jiang Z, Zhang X, Gu A. Gut microbiota promotes cholesterol gallstone formation by modulating bile acid composition and biliary cholesterol secretion. Nat Commun 2022; 13(1): 252.
5. Dwivedi M, Dwivedi M, Moitra M, Sanyal S. The implication of gall stones in gallbladder cancer and recent updates on its epidemiology. In: Gallstone Formation, Diagnosis, Treatment and Prevention. Elsevier, 2024, p. 207‒222.
6. Hall L, Halle-Smith J, Evans R, Toogood G, Wiggins T, Markar SR, Kapoulas S, Super P, Tucker O, McKay SC. Ursodeoxycholic acid in the management of symptomatic gallstone disease: Systematic review and clinician survey. BJS Open 2023; 7(2): zrac152.
7. Mulliri A, Menahem B, Alves A, Dupont B. Ursodeoxycholic acid for the prevention of gallstones and subsequent cholecystectomy after bariatric surgery: A meta-analysis of randomized controlled trials. J Gastroenterol 2022; 57(8): 529‒539.
8. Sharma A, Shanti H, Nageswaran H, Best LM, Patel AG. Role of ursodeoxycholic acid in the prevention of gallstones formation in bariatric patients ‒ a systematic review and meta-analysis of randomised trials. Obes Surg 2023; 33(12): 4115‒4124.
9. Payne T, Appleby M, Buckley E, van Gelder LMA, Mullish BH, Sassani M, Dunning MJ, Hernandez D, Scholz SW, McNeill A, Libri V, Moll S, Marchesi JR, Taylor R, Su L, Mazzà C, Jenkins TM, Foltynie T, Bandmann O. A double-blind, randomized, placebo-controlled trial of ursodeoxycholic acid (UDCA) in Parkinson’s disease. Mov Disord 2023; 38(8): 1493‒1502.
10. Portincasa P, Di Ciaula A, Bonfrate L, Stella A, Garruti G, Lamont JT. Metabolic dysfunction-associated gallstone disease: Expecting more from critical care manifestations. Intern Emerg Med 2023; 18(7): 1897‒1918.
11. Wang HH, Portincasa P, Liu M, Wang DQH. Effects of biliary phospholipids on cholesterol crystallization and growth in gallstone formation. Adv Ther 2023; 40(3): 743‒768.
12. Lange AH, Pedersen MG, Ellegaard AM, Nerild HH, Brønden A, Sonne DP, Knop FK. The bile-gut axis and metabolic consequences of cholecystectomy. Eur J Endocrinol 2024; 190(4): R1‒R9.
13. Fiorucci S, Urbani G, Di Giorgio C, Biagioli M, Distrutti E. Bile acids-based therapies for primary sclerosing cholangitis: Current landscape and future developments. Cells 2024; 13(19): 1650.
14. Sperna Weiland CJ, Verschoor EC, Poen AC, Smeets XJMN, Venneman NG, Bhalla A, Witteman BJM, Timmerhuis HC, Umans DS, van Hooft JE, Bruno MJ, Fockens P, Verdonk RC, Drenth JPH, van Geenen EJM; Dutch Pancreatitis Study Group. Suspected common bile duct stones: Reduction of unnecessary ERCP by pre-procedural imaging and timing of ERCP. Surg Endosc 2023; 37(2): 1194‒1202.
15. Sebghatollahi V, Parsa M, Minakari M, Azadbakht S. A clinician’s guide to gallstones and common bile duct (CBD): A study protocol for a systematic review and evidence-based recommendations. Health Sci Rep 2023; 6(9): e1555.
16. Song P, Zhang X, Feng W, Xu W, Wu C, Xie S, Yu S, Fu R. Biological synthesis of ursodeoxycholic acid. Front Microbiol 2023; 14: 1140662.
17. Robles-Díaz M, Nezic L, Vujic-Aleksic V, Björnsson ES. Role of ursodeoxycholic acid in treating and preventing idiosyncratic drug-induced liver injury. A systematic review. Front Pharmacol 2021; 12: 744488.
18. Sarcognato S, Sacchi D, Grillo F, Cazzagon N, Fabris L, Cadamuro M, Cataldo I, Covelli C, Mangia A, Guido M. Autoimmune biliary diseases: Primary biliary cholangitis and primary sclerosing cholangitis. Pathologica 2021; 113(3): 170‒184.